Description
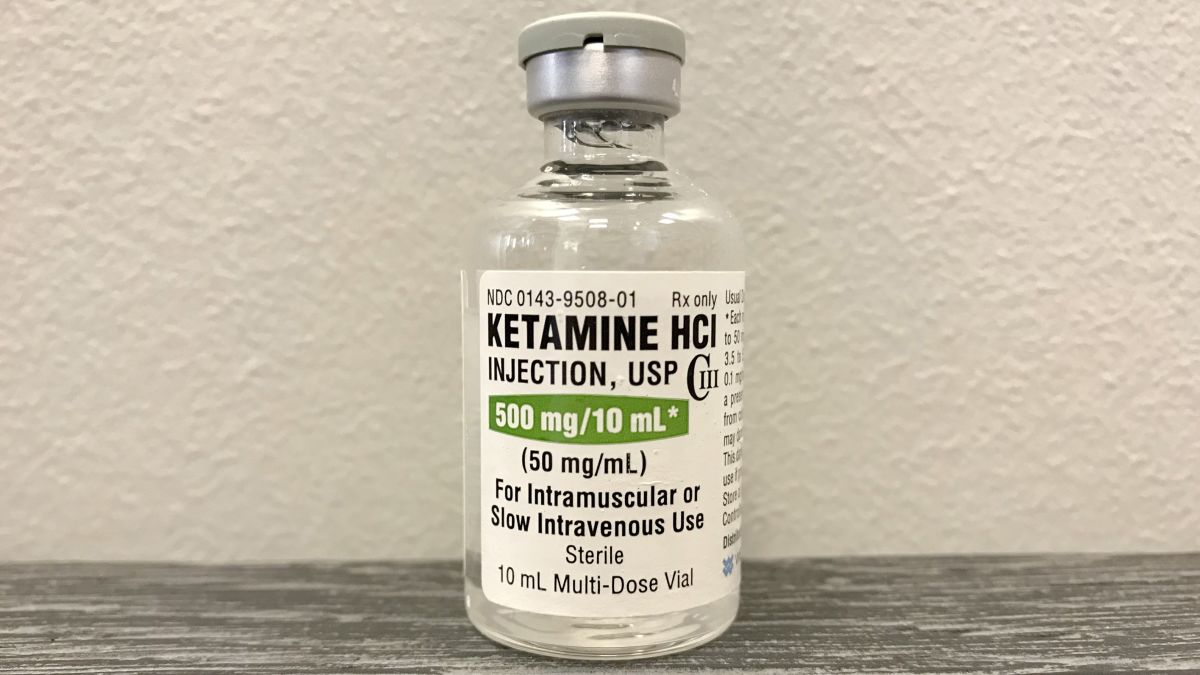
Die Bereitstellung hochreiner, gebrauchsfertiger Substanzen ist für die Reproduzierbarkeit moderner neurowissenschaftlicher Studien unerlässlich. Möchten Sie Ketamin HCL für Forschung kaufen? Unsere Ketamine HCL-formulation as Sterile liquid solution is exclusively for qualified Scientific studies in strictly controlled laboratory facilities. The concentrations of 50 mg/ml and 100 mg/ml eliminieren signifikante Fehlerquellen bei der Dosierung. Dies gewährleistet die Konsistenz über alle Versuchsreihen hinweg. Als controlled substance unterliegt der Erwerb und die Handhabung strengsten internen und ethischen Protokollen. Deren Einhaltung ist für jede Forschungseinrichtung zwingend.
Was ist Ketamin HCL?
Das Verständnis der chemischen Basis und die Vorteile der flüssigen Darreichungsform sind entscheidend für präzise pharmakologische und zelluläre Assays. Hier bieten wir nur Top Qualität Ketamin HCL to.
Chemische Struktur und Hydrochlorid-Form
Ketamine is a chiral molecule. Es wird oft als Racemat verwendet, manchmal die enantiomerenreine S-form (esketamine). Es enthält eine Cyclohexane group and a Amino group, verantwortlich für seine pharmakologische Aktivität. Die HCL salt-form (hydrochloride) is a Water-soluble salt. Dies ist ideal für die Herstellung von Lösungen. Die Form ist unerlässlich für die exakte Dosierung in flüssigen Forschungssystemen. Sie erlaubt einfache Inkorporation in Zellkulturmedien oder physiologische Puffer.
Warum Ketamin HCL Online Kaufen bei chemgramx?
Die Formulierung als sterile Liquid-Lösung bietet erhebliche Vorteile gegenüber Pulver. Besonders in der präklinischen und zellulären Forschung muss Kontamination vermieden werden. Bei uns finden Sie das Bestes Ketamin HCL Verfügbar. Kaufen Sie Ihre Forschungschemikalien sicher bei chemgramx.
- Sterility: The solution is produced under clean room conditions Filtered several times. Dies garantiert Sterility and Absence of pyrogens. Sie kann direkt in zellulären oder tierischen Modellen verwendet werden, ohne Kreuzkontamination oder inflammatorische Reaktionen zu riskieren.
- Precision: The ready-to-use concentrations 50 mg/ml and 100 mg/ml eliminieren Fehlerquellen beim manuellen Einwiegen und Lösen des Pulvers. Dies gewährleistet die Consistency of dosing über alle Versuche und optimiert die Reproducibility Ihrer Forschung.
- Solvent: High purity is used as a solvent, Pyrogen-free water. Ein physiologisch optimierter pH-Wert erhöht die Stabilität der Formulierung und verbessert die Zellverträglichkeit.
Wirkmechanismus und Wissenschaftliche Anwendungsbereiche
Ketamin ist primär für seinen einzigartigen und schnellen Wirkmechanismus bekannt. Dies macht es zu einem zentralen Forschungsobjekt in der Neurobiologie.
NMDA receptor antagonism
Ketamine acts as non-competitive antagonist on NMDA receptor (N-Methyl-D-Aspartat-Rezeptor). Es blockiert den Ionenkanal dieses ionotropen Glutamat-Rezeptors. Diese Blockade reduziert die exzitatorische synaptische Übertragung im zentralen Nervensystem. Diese Interaktion ist der Grundstein für neurowissenschaftliche Studien. Sie untersuchen die Rolle des Glutamate system bei Lernprozessen, Synaptischer Plastizität, Neurodegeneration und affektiven Störungen. Produkte wie Mexedrone können ebenfalls für verwandte Forschungen relevant sein.
Spezialisierte Forschungsfelder
The sterile ketamine solution is used in various highly specialized fields of research:
- Psychiatric research: Ketamin hat enormes Forschungspotenzial in Depression and therapy studies. It serves as a model to Rapid onset of antidepressant effect und molekulare Mechanismen (z. B. synaptische Reparatur, erhöhte Neuroplastizität) zu untersuchen.
- Pain research: In the Analgesia wird Ketamin erforscht. Ziel ist es, analgetische Wirkmechanismen zu verstehen, besonders bei Chronic and neuropathic pain conditions. The focus is on blocking central sensitization pathways.
- Biochemical analyses: The sterile solution is used for precise Receptor binding studies verwendet. Sie bestimmt die Affinität zu anderen Monoamin-Rezeptoren oder Transportproteinen.
- Preclinical models: Die sterile Injektionslösung ist für In-vivo-Studien in Tiermodellen unerlässlich. Ethische und interne Standards sind dabei stets zu gewährleisten.
Qualitätssicherung und Laborsicherheit
The handling of Ketamine HCL erfordert aufgrund seiner Klassifizierung als kontrollierte Substanz strengste Kontrollen. Eine lückenlose Dokumentation ist ebenso erforderlich. Wir bieten Ihnen stets High purity for sale.
Qualität und Dokumentation
- Degree of purity: The purity of the active ingredient ≥ 99% is characterized by HPLC analysis bestätigt. Jede Charge ist vollständig dokumentiert und rückverfolgbar. Dies garantiert unsere Research quality for sale.
- Sterility guarantee: Filtration und Produktion unter Reinraumbedingungen gewährleisten die Sterilität und Abwesenheit von Pyrogenen. Diese Dokumente sind für eine GLP-konforme Berichterstattung zwingend erforderlich.
Sicherer Umgang im Labor
Dieses Produkt ist strengstens not for human consumption bestimmt. Es ist auch nicht für nicht-analytische oder klinische Zwecke vorgesehen.
- Exclusively for research: The purchase is only for documented research facilities möglich. Dazu gehören Universitäten und Pharmaunternehmen mit offiziellem Nachweis. Eine Description of the research project ist ebenfalls notwendig. Lieferungen an private Adressen sind ausgeschlossen.
- Laboratory safety: The work must exclusively by qualified personnel erfolgen. Dies geschieht unter einem Chemical fume cupboard und mit steriler Schutzkleidung. Die Entsorgung hat nach geltenden internen Richtlinien für kontrollierte Substanzen zu erfolgen.
Fazit: Ketamin HCL Online Bestellen
Zusammenfassend bieten die sterilen Ketamine HCL Lösungen die ideale Basis für zuverlässige und reproduzierbare Forschung. Dies gilt in der Neurobiologie und Psychiatrie. Wenn Sie Ketamin HCL Online Bestellen möchten, setzen Sie auf unser hochwertiges Produkt. Ein Secure purchase für Ihre qualifizierte Forschung ist stets gewährleistet.




Reviews
There are no reviews yet.